Abstract
TC10 cell counter can replace the LDH assay, to measure both the viability of cell suspensions and the cytotoxic effects of various compounds.
Introduction
Determination of cell viability is an important issue when working with cellular models. Various assays, based on morphological (e.g., electron microscopy) or biochemical (e.g., caspase activation) characteristics of dying or dead cells, have been developed to assess cell death–related parameters. Among these assays, those measuring membrane integrity are the most commonly used, even if they cannot discriminate between the different known forms of cell death. Loss of plasma membrane integrity is considered a point-of-no-return while the occurrence of other biochemical events does not necessarily mean that cell death will ensue (Kroemer et al. 2009). The loss of membrane integrity can be measured by using exclusion dyes (i.e., dyes, such as trypan blue, that cannot enter healthy cells but that are taken up by cells with permeabilized plasma membranes) or by measuring the activity of cytosolic enzymes (e.g. LDH) released in the culture supernatants after rupture of the plasma membrane.
Here we present the results of a comparison study between the TC10 cell counter and the classical LDH assay to measure cell viability in suspension cells treated with two different toxic agents.
Materials and Methods
K562 cells were maintained in RPMI medium supplemented with 10% fetal calf serum, streptomycin (100 µg/ml) and penicillin (100 IU/ml) at 37°C in a 95% air/5% CO2 atmosphere with 100% humidity. For the experiments, K562 cells were incubated at a concentration of 1 x 106 cells/ml.
Cell viability was estimated by using the TC10 automated cell counter (Bio-Rad Laboratories, Inc.), following manufacturer’s instructions. An aliquot of the cell suspension (10 µl) was added to 10 µl of 0.4% trypan blue solution (Bio-Rad) in a test tube and mixed by gently pipetting up and down several times. Ten microliters were then transferred into the counting chamber of a TC10 counting slide and analyzed using the TC10 cell counter.
LDH activity was measured according to the procedure of Wroblewski and Ladue (1955), both in the culture medium and in the cell pellet obtained after centrifugation. Briefly, an aliquot of cell suspension (1 ml) was centrifuged at 1,000 x g for 1 min at 4°C. Five hundred microliters of cell-free supernatant were saved without disturbing the cell pellet. The remaining supernatant was discarded and the pellet was suspended in 1 ml of ice-cold phosphate buffer (pH 7.4). Both resuspended pellet and supernatant samples were treated by addition of Triton X-100 (2% final concentration) to lyse cells and diluted by three-fold and two-fold, respectively, with distilled water. Fifty microliters of these solutions were placed in 2.75 ml of phosphate buffer, in the presence of NADH (120 µM). Samples were heated at 37°C for 20 min and the LDH reaction was started by the addition of pyruvate (600 µM). NADH conversion to NAD+ was monitored spectrophotometrically at 340 nm. Cell viability was measured by calculating the ratio of release LDH activity to total activity. Results are expressed as 100% minus the ratio of released activity to the total activity.
Results and Discussion
K562 cells, an erythroleukemia cell line, were treated with 25 µM of doxorubicin, an anthracyclin antibiotic commonly used to treat several types of cancers and two concentrations of menadione (50 and 200 µM), a redox cycling agent that induces an oxidative stress. Twenty four hours after treatment, cell viability was determined by measuring trypan blue exclusion using the TC10 cell counter and by measuring LDH activity. The two methods gave very similar viability results for the doxorubicin treatment experiment, showing a 40% decrease of cell viability. Similar cell viability results were also obtained with the two methods for the menadione treatment. However, cell viability reported by the TC10 cell counter was lower than that reported by the LDH assay (40% and 57%, for the LDH assay and the TC10 cell counter, respectively).
Based on these data, we propose that the TC10 cell counter can advantageously replace the LDH assay, both to measure the viability of cell suspensions and the cytotoxic effects of various compounds. Cell counting with the TC10 cell counter is faster than performing a long and time-consuming biochemical assay (a few minutes instead of several hours) and it avoids the classical pitfalls encountered with the LDH assay (e.g., natural degradation of the enzyme, influence of the pH, inhibition by specific components of the culture medium).
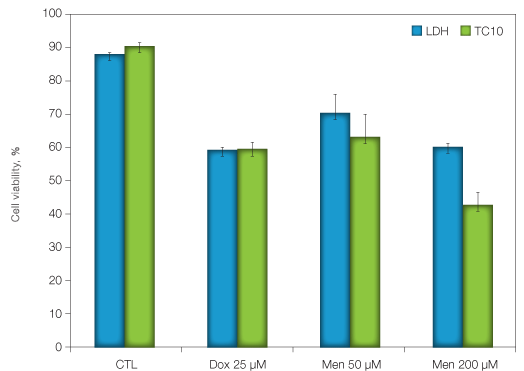
Fig. 1. Determination of cell viability after exposure to different toxic agents. K562 cells were incubated at a density of 1 x 106 cells/ml and exposed to the anthracyclin antibiotic doxorubicin or the pro-oxidant menadione for 24 hours. Cell viability was determined as indicated in the Materials and Methods section.
Acknowledgements
The authors are grateful to Isabelle Blave and Véronique Allaeys for their excellent technical assistance.
References
Kroemer G et al. (2009). Classification of cell death: Recommendations of the nomenclature committee on cell death 2009. Cell Death Differ 16, 3-11.
Wroblewski F and Ladue JS (1955). Lactic dehydrogenase activity in blood. Proc Soc Exp Biol Med 90, 210-213.

